Module 2: Refinery Processes & Units
Reforming and Isomerization: Upgrading Naphtha
Straight-run naphtha does not have a high enough octane rating to be used effectively in gasoline blending. Refiners use two main processes to increase its octane: reforming and isomerization. The general principle behind both is rearranging straight-chain hydrocarbons (n-paraffins) into branched and iso-paraffins, which burn more cleanly and have higher octane.
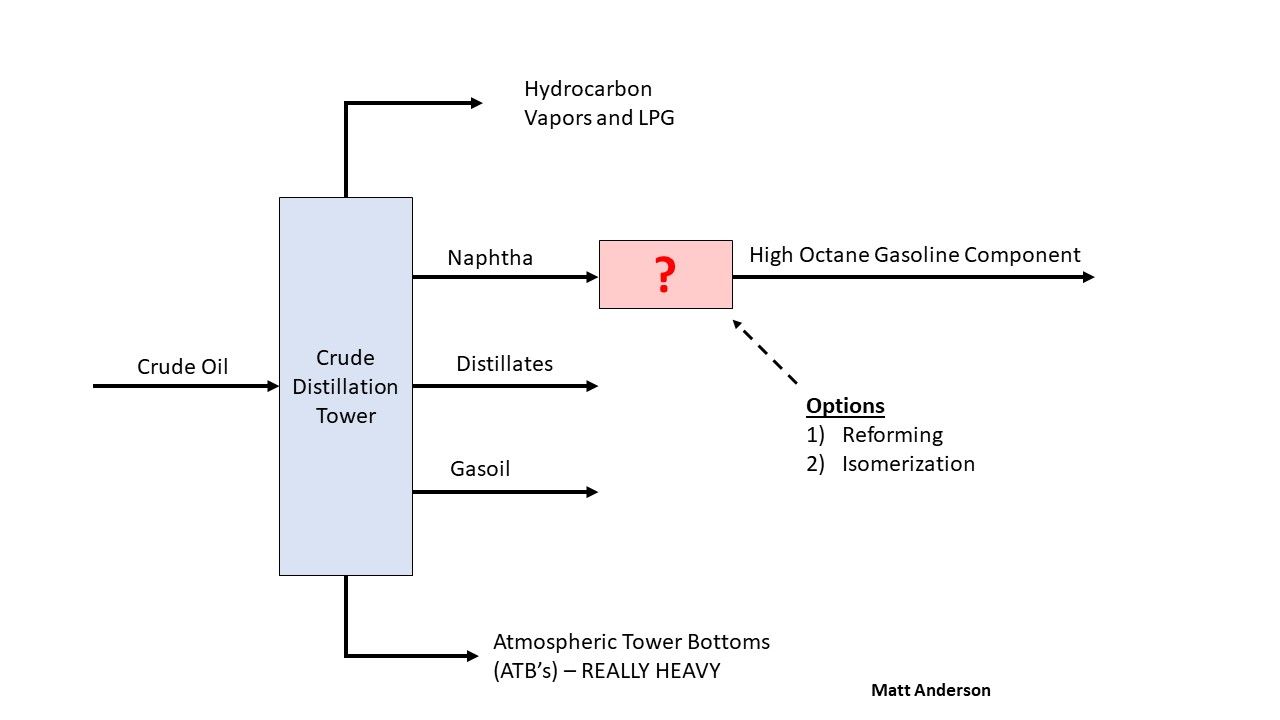
Reforming
The product of reforming is called "reformate". Octane increases through the production of isoparaffins and aromatics. Hydrogen is produced as a byproduct and is used elsewhere in the refinery, such as in hydrotreating. The feed must be hydrotreated first, because sulfur will poison the catalyst. This also means reformate has very low sulfur, which is beneficial for gasoline blending. UOP commercialized reforming and built the first unit, called a "Platformer", in 1949.
Isomerization
The product of isomerization is called "isomerate". The feed is typically C5 and C6 molecules, which are lighter than reformer feed. Straight-chain pentane (n-pentane) has a Research Octane Number (RON) of about 60, while isopentane has a RON of 90. The process uses a platinum catalyst, consumes hydrogen, and saturates any olefins present in the feed. Like reforming, the feed must be hydrotreated to remove sulfur before entering the unit. "Penex" is the most common commercial isomerization technology for C5 and C6 molecules, developed and licensed by UOP.
Why It Matters
Upgrading naphtha turns a low-value intermediate into a high-octane gasoline blendstock. Without these processes, refiners would struggle to meet finished gasoline octane specs using straight-run material alone.
References
Handbook of Petroleum Refining Processes, 4th Ed., Meyers