Module 2: Refinery Processes & Units
The Bleach Cycle: Salt, Chlorine, Water
Bleach is a dilute solution of sodium hypochlorite and water. Refineries use it in wastewater treatment because it is an effective oxidizer. For context, Clorox disinfecting bleach is about 7% sodium hypochlorite and 93% water.
Where the Chlorine Comes From
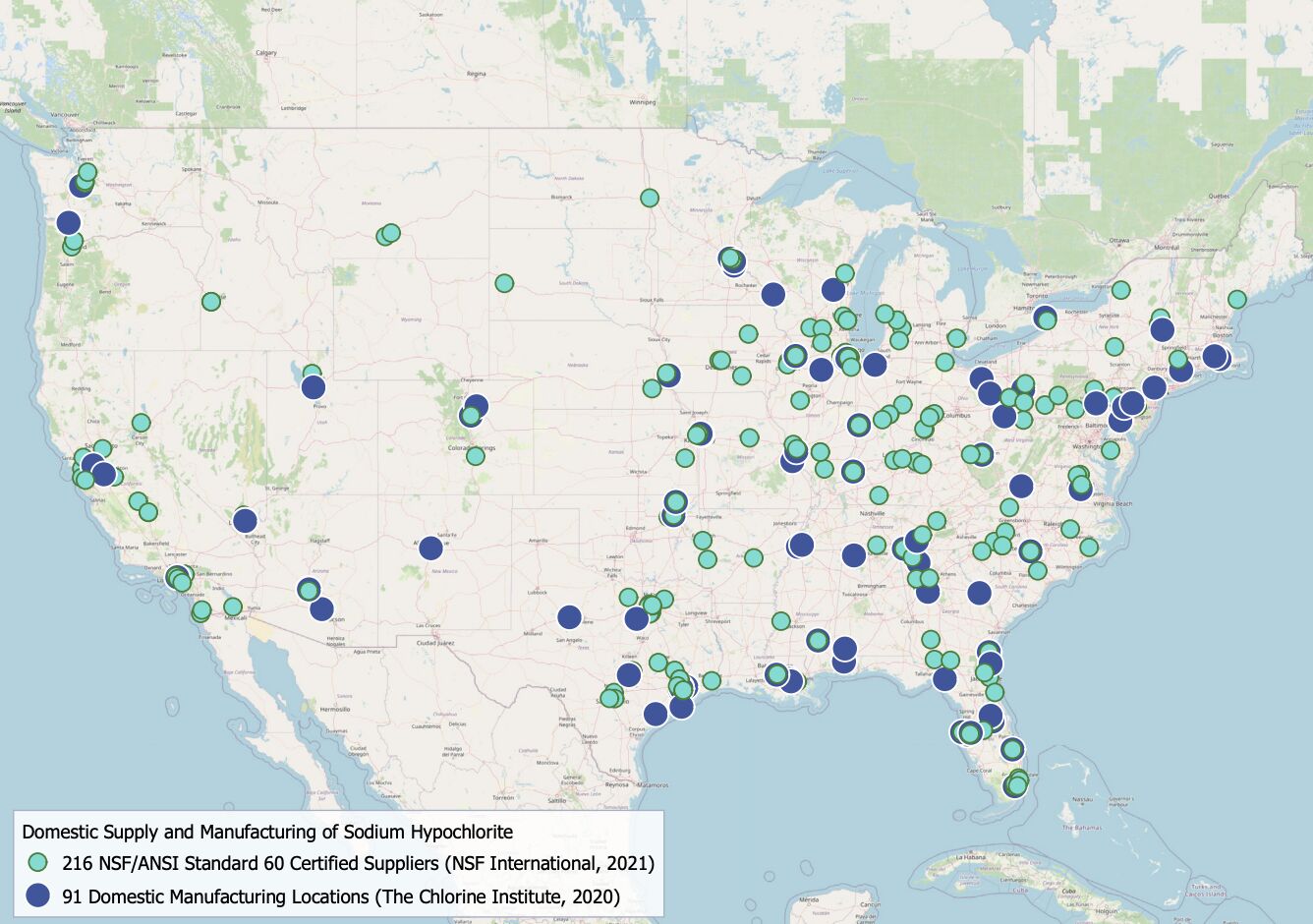
The chlorine in bleach mostly comes from the ocean. Brine, which is concentrated salt water, is used to produce chlorine gas through electrolysis. Chlorine gas and sodium hydroxide are then combined to produce sodium hypochlorite. In 2015, the United States consumed roughly 962 million kg of bleach. As of 2020, there were 91 bleach manufacturers in the country.
Bleach Has a Short Shelf Life
Bleach decomposes over time. Its shelf life is approximately one month. The sodium hypochlorite gradually reacts and breaks down into salt and water, the same materials it started from.
It Is a Cycle
Chlorine gas is produced from salt water, used to make bleach, and the bleach eventually decomposes back into salt and water. The diagram below, from the EPA, illustrates this cycle.