Module 1: Hydrocarbon Chemistry, Feedstocks & Products
Straight Chains and Branches: A Look at Paraffin Types in Refining
Normal paraffins and Isoparaffins are both hydrocarbons, but a few of their properties diverge significantly.
"Normal" means straight chain while "iso" signifies branching. The diagram included with this post includes a few examples.
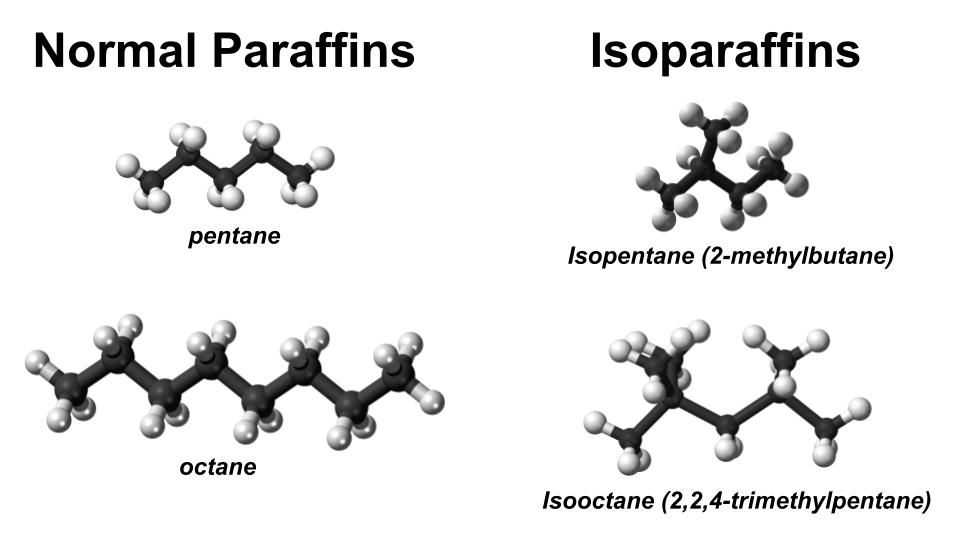
Isoparaffins Have Much Higher Octane
➡️ The octane number (what you see at the gas station) of isooctane is 100! In fact, the gasoline octane scale is defined by isooctane.
➡️ The octane number of normal octane is negative! The straight-chain structure of normal octane makes it more likely to combust prematurely, which causes knocking.
Normal Paraffins Have a Higher Pour Point
➡️ The pour point is the minimum temperature required for the hydrocarbon to flow. For example, if a gas oil pour point is 60 degF, then the hydrocarbon will start to solidify and stop flowing when the temperature goes below 60 degF.
➡️ High pour point is a problem when it gets close to ambient temperature. If the ambient temperature cools while pipes are filled with high pour point gas oil, then the lines could plug.
➡️ Isoparaffins have lower pour point because the branched structure makes the formation of crystal structures more difficult. Therefore, the hydrocarbon is still able to flow at lower temperatures.
Why Is This Helpful at All?
✅ Maybe it isn't...but if you work in an oil refinery, it's valuable to understand different ways to classify hydrocarbons and their properties.
✅ The situation could be that you are offered a high pour point gas oil at a discount to purchase. When you hear "high pour point" you can gain some insight into the fact that it probably has a good amount of normal paraffins. Fortunately, your site has a hydrocracker, and you know that one of the reactions that occurs in a hydrocracker is isomerization. Through isomerization, normal paraffins can be converted into isoparaffins.