Module 1: Hydrocarbon Chemistry, Feedstocks & Products
Hydrogen Content: Gasoline vs. Middle Distillates
Hydrogen content is an important parameter for understanding gasoline, jet fuel, and diesel. Each fuel has different requirements, and understanding why helps explain some of the refinery's most important processes.
The Stangeland Diagram
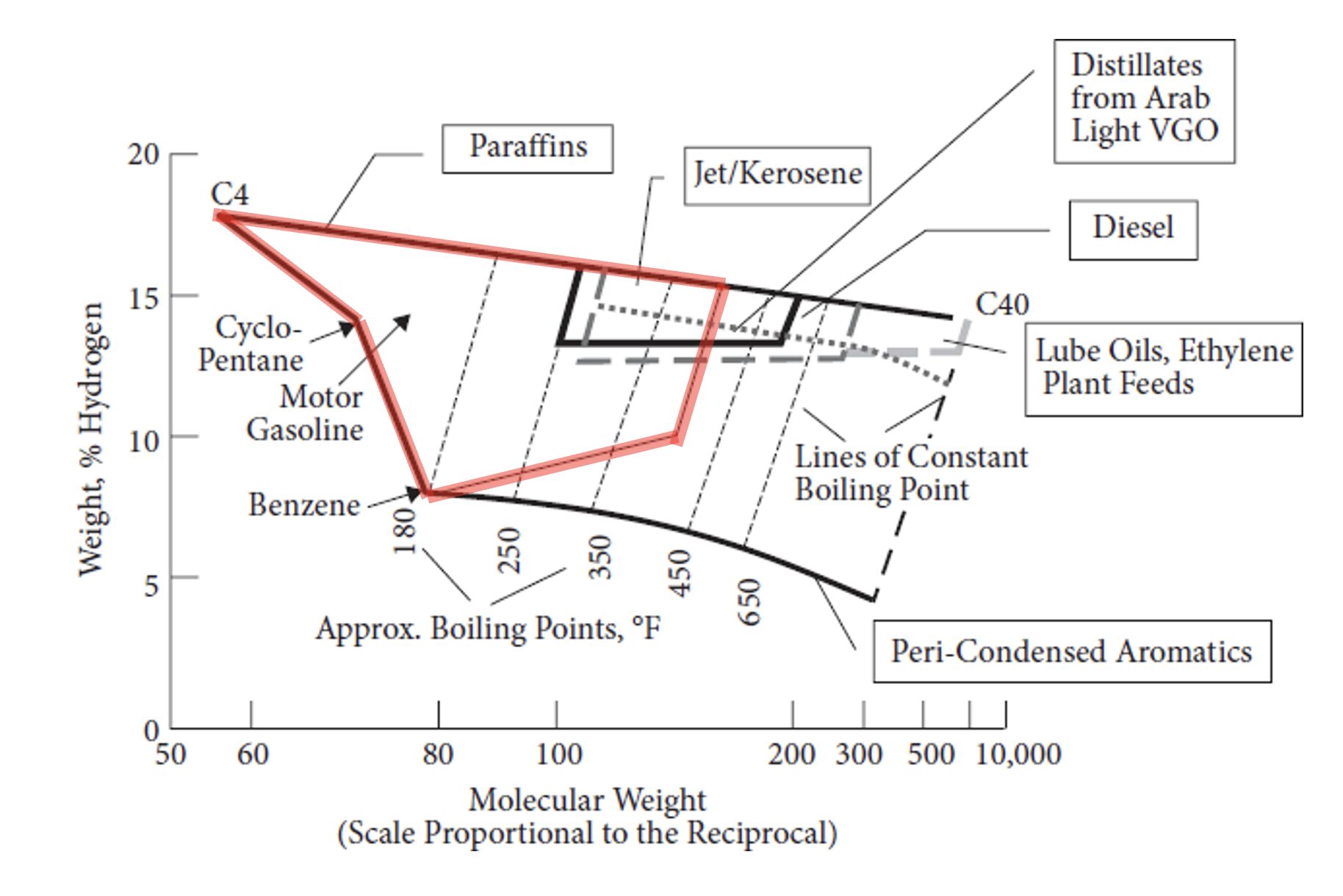
The Stangeland Diagram relates the hydrogen weight percentage of hydrocarbon molecules to their molecular weight. It can be used to check acceptable hydrogen percentages for different fuel products.
Each Fuel Has a Different Combustion Spec
Gasoline, jet fuel, and diesel are each evaluated against a different combustion specification. Gasoline is rated by octane. Jet fuel is rated by smoke point. Diesel is rated by cetane.
Why Jet Fuel and Diesel Need High Hydrogen
Jet fuel and diesel (known as "middle distillates") have a much narrower range of acceptable hydrogen content. Heavy aromatic compounds prevent these products from meeting their cetane and smoke point specs. The aromatics must be removed: saturated with hydrogen and possibly cracked, to meet specs. This is why the Stangeland diagram shows the jet fuel and diesel regions bordering the paraffin line at the top, which represents the maximum possible hydrogen weight percentage.
Gasoline Is More Flexible
For gasoline, the range of acceptable hydrogen percentage is much larger. Aromatics, which are hydrogen-deficient, actually have high octane values. So a gasoline blender can tolerate more aromatic content than a jet fuel or diesel producer can.
How You Add Hydrogen and Remove Aromatics
Hydrocracking and hydrotreating are the two main processes. Both add hydrogen and saturate aromatics. Hydrocracking also breaks molecular bonds and isomerizes molecules to produce branched structures.