Module 2: Refinery Processes & Units
Sloppy Cuts: When Separation Isn't Perfect
Gasoline, jet fuel, and diesel are all hydrocarbons. The main difference is their molecule size, which determines the temperature range that they boil (called distillation profile). Larger molecules generally boil at higher temperature.
Refiners have a limited ability to shift molecules between the three fuels. This helps to respond to demand changes.
What Is a Cut Point?
"Cut Point" is a key term for understanding this concept. A formal definition for cut point is "temperature that represents the limit of a distillate fraction". [1]
This is how I think about cut point:
If separation between two products was perfect, then the cut point would be the maximum boiling point (called end point) for the lighter product and the minimum boiling point (called initial boiling point) for the heavier product.
Sloppy Cuts and Swing Molecules
However, separation is never perfect. When separation is especially poor, this is called having "sloppy cuts".
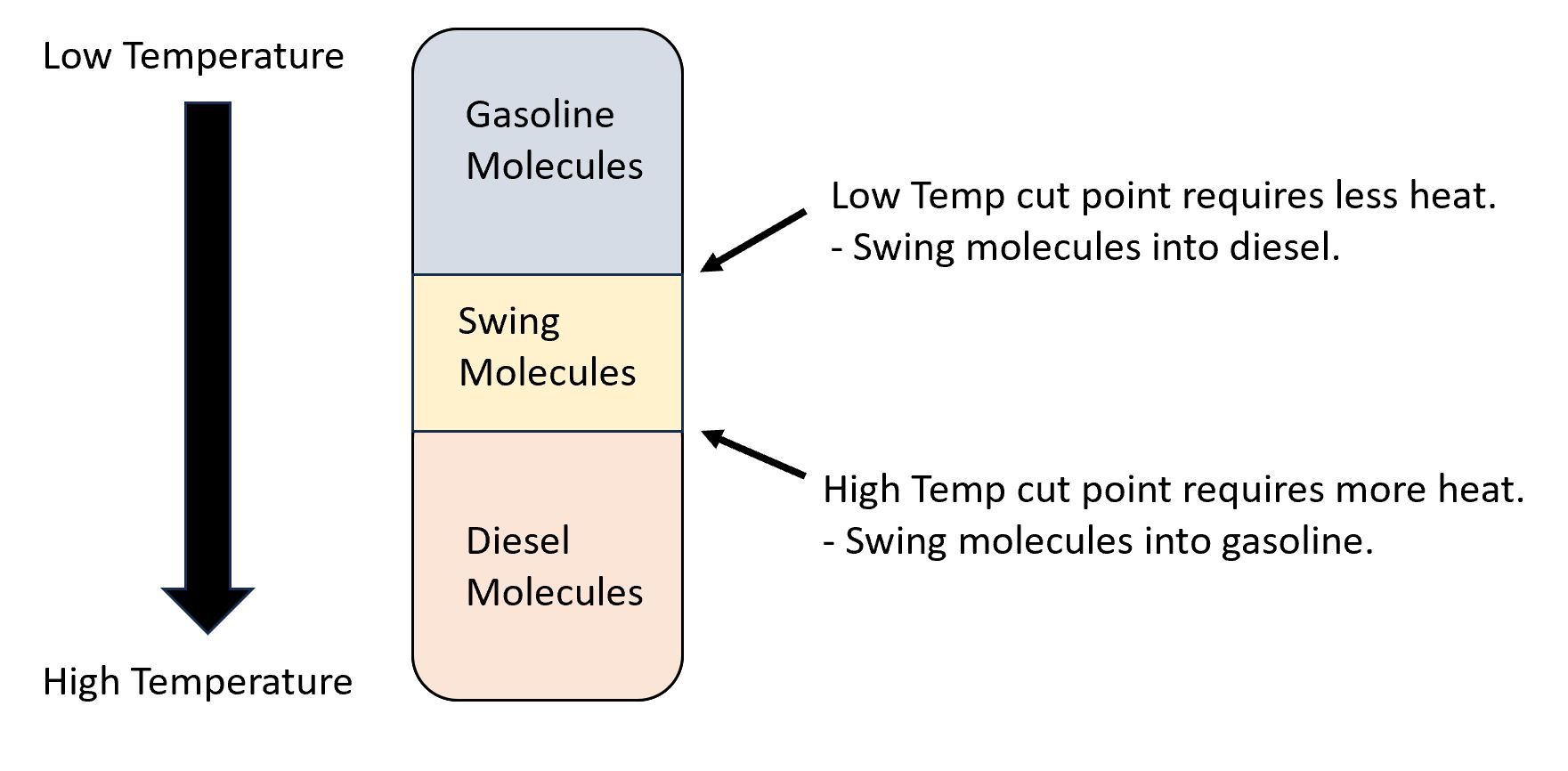
Imperfect separation results in overlap between products. The lightest molecules in diesel could be gasoline. The heaviest gasoline molecules could be diesel. These borderline molecules are sometimes called "swing cut".
Adjusting Cut Points with Heat
Refiners can adjust the volume of products by adding or removing heat to distillation columns. This changes the product compositions. When heat is added, lighter molecules are driven further up the tower. Lab tests are run on products to determine exact distillation profiles and heat is adjusted.
In the theoretic separation between gasoline and diesel, adding heat would force more hydrocarbon molecules into gasoline. The result would be a higher gasoline/diesel cut point. Reducing heat would compel molecules to drop back into diesel and lower the gasoline/diesel cut point.
A higher cut point results in higher yield of the lighter product.