Module 2: Refinery Processes & Units
Hydrotreating: Removing Sulfur with Hydrogen
Hydrotreating is a refinery process that removes objectionable materials such as sulfur, nitrogen, and metals. It also saturates olefins and aromatics. Meeting modern gasoline and diesel standards would not be possible without it. A simple way to think about hydrotreating is swapping contaminants with hydrogen.
Three Types of Feed
Hydrotreating technology is divided roughly by feed type. The three main categories are naphtha, distillate, and gas oil. Naphtha is the lightest and gas oil is the heaviest. As the feed gets heavier, you can generally expect higher hydrogen consumption per barrel, higher reactor temperature, and higher reactor pressure.
The Handbook of Petroleum Refining Processes provides these rough estimates for hydrogen consumption in Standard Cubic Feet per Barrel of feed: naphtha requires 40 to 500 SCF/BBL, distillate requires 130 to 580 SCF/BBL, and gas oil requires 250 to 760 SCF/BBL.
Sterically Hindered Sulfur
Larger molecules, such as those found in gas oil, can contain sulfur species that are particularly difficult to remove. Dibenzothiophene is a common example. You will hear the phrase "sterically hindered" used to describe these molecules. It means the sulfur atom is well protected by the surrounding molecular structure, making it physically difficult for the hydrogenation reaction to reach it.
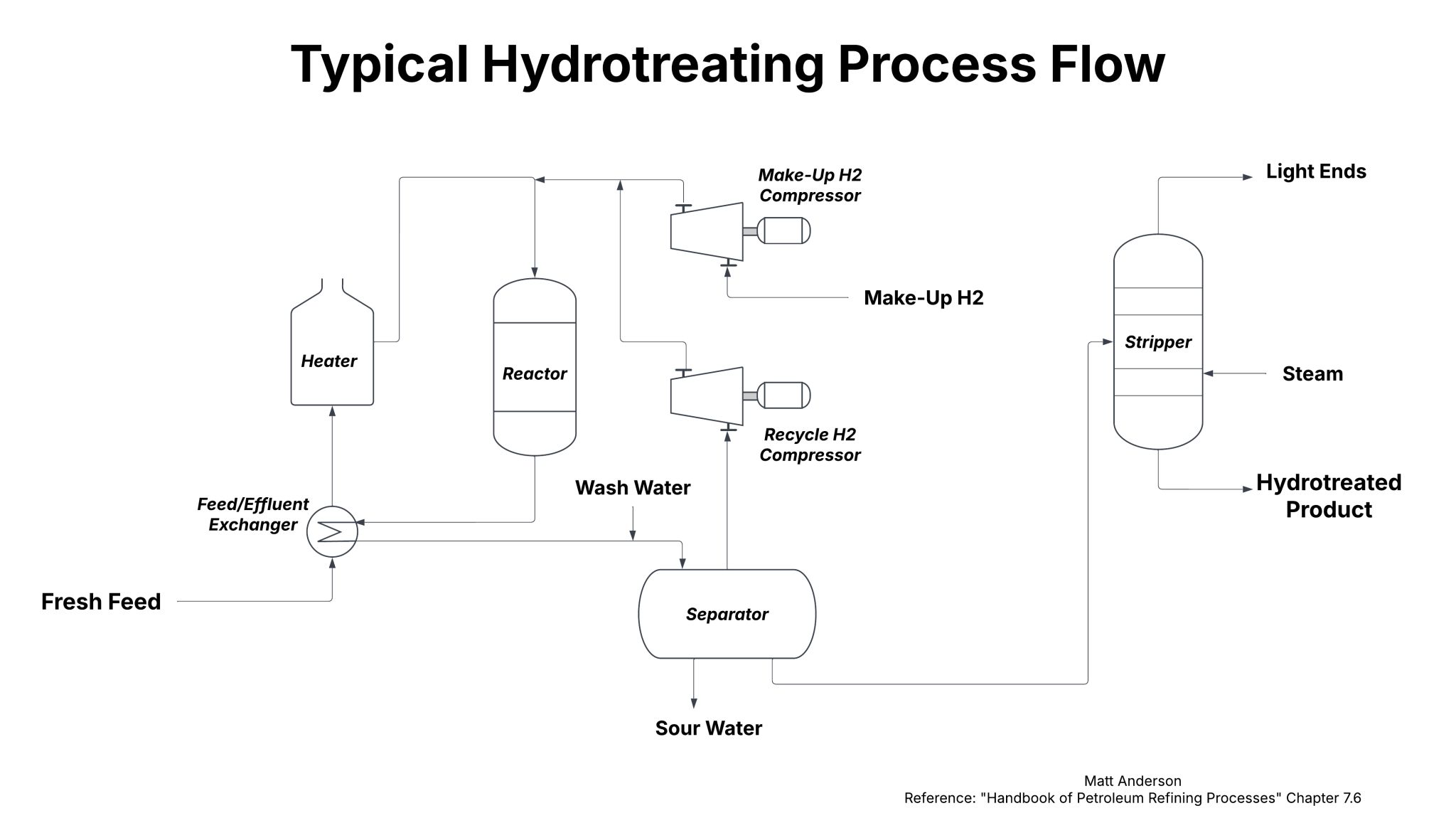
Process Flow
Most hydrotreating units follow a similar arrangement. Fresh feed is heated and combined with hydrogen before contacting the catalyst in a reactor. After the reactor, gas and liquid streams are separated. Hydrogen is recycled back to the front of the unit, while the liquid stream goes through further separation in a trayed column called a stripper.