Module 3: Equipment, Inspection & Maintenance
How Ammonium Salts Form in Refineries
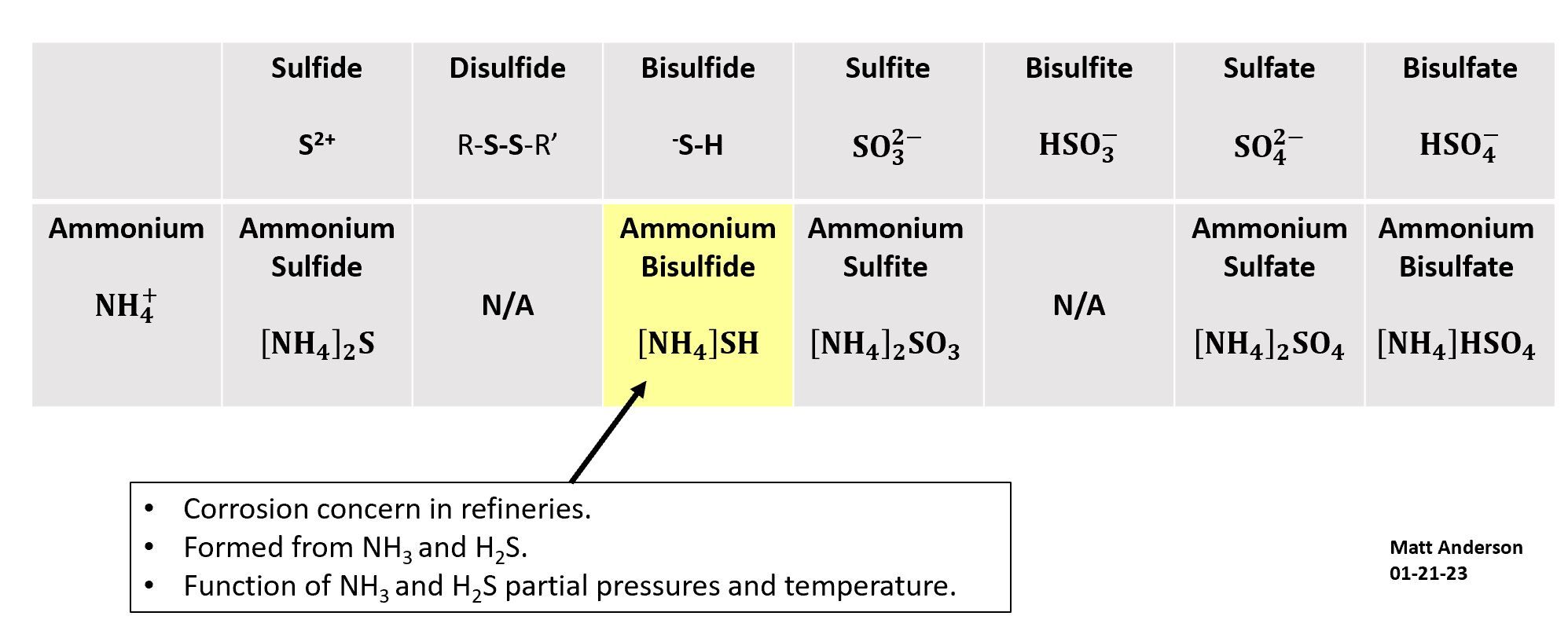
Ammonium salt formation in refineries causes two problems: corrosion and fouling. The two most common ammonium salts are ammonium bisulfide and ammonium chloride.
How Salts Form
Salts form from a reaction between an acid and a base. Ammonium bisulfide is formed when ammonia (NH3) acts as the base and hydrogen sulfide (H2S) acts as the acid. Both compounds are present in the vapor phase, and the reaction produces ammonium bisulfide as a water-soluble solid. Ammonium chloride forms by a similar mechanism, with hydrogen chloride serving as the acid.
What Controls the Rate of Formation
The rate of ammonium bisulfide formation depends on two factors: the partial pressures of NH3 and H2S, and temperature. Lower temperatures favor more salt formation. This is why ammonium salt deposition is most likely to occur at cooler points in a process unit, such as heat exchanger bundles, where the combination of high partial pressures and low temperature creates the right conditions.
The Salt Family
The table below shows sulfur anions paired with the ammonium cation to illustrate the range of salts that can form. Ammonium bisulfide is the one most commonly cited as problematic in refinery operations. The prefix "bi" in chemistry denotes one hydrogen atom, so ammonium bisulfide contains one hydrogen in its anion.